
Introduction
Pick the wrong surface finishing method and you're looking at rework costs, finish mismatches between components, or a coating that fails in the field. PVD and electroplating are both proven options — used across cosmetic packaging, automotive parts, medical devices, and consumer electronics — but they work through fundamentally different processes and suit different applications.
The right choice comes down to your substrate, performance requirements, and production volume. For manufacturers running multi-component products across cosmetics, automotive, or electronics, understanding these trade-offs directly affects finish consistency and total production cost.
TL;DR
- PVD deposits material in a vacuum chamber via vaporization; electroplating uses an electrolytic chemical bath
- PVD produces harder, thinner, more adhesive coatings; electroplating builds thicker layers with weaker bonding
- PVD suits high-durability, tight-tolerance applications; electroplating fits decorative finishes and precious metal deposition
- PVD works on a wider material range — including engineering plastics — while electroplating requires conductive surfaces
- The two methods can combine—PVD as base layer, electroplating for precious metal aesthetics
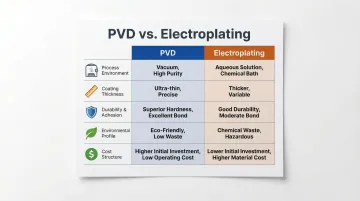
PVD vs. Electroplating: Quick Comparison
Here's how PVD and electroplating compare across the five factors that most often drive material selection decisions.
| Factor | PVD | Electroplating |
|---|---|---|
| Process Environment | Vacuum chamber — solid material vaporizes and condenses onto substrate | Chemical bath — electrical current deposits metal ions onto submerged parts |
| Coating Thickness | 2–5 microns — preserves tight tolerances | 5–500 microns (hard chrome) — risks edge build-up and tolerance loss |
| Durability & Adhesion | Atomic-level bond via ion bombardment (50–100 eV); scratch load >50 N | Surface-level bond; pull-off strength 2–6.88 MPa — prone to peeling on hard substrates |
| Environmental Profile | No hazardous chemical waste | Requires EPA compliance (40 CFR 413/433) for wastewater and chemical bath management |
| Cost Structure | Higher upfront equipment cost; no ongoing disposal fees — total cost of ownership is competitive at scale | Lower upfront cost; ongoing chemical disposal and compliance costs add up |
What is PVD Coating?
Physical Vapor Deposition (PVD) is a vacuum-based process where solid coating materials—typically titanium, zirconium, or chromium—are vaporized and then condense onto a substrate, forming thin, dense, highly adherent films. This process creates molecular-level bonding rather than surface deposits.
That bonding strength comes from a combination of high-energy ion bombardment, in situ sputter cleaning of the substrate surface, and simultaneous deposition with substrate sputtering. Together, these remove oxide layers and penetrate the substrate lattice—creating a graded interface rather than a surface layer that can peel or chip.
PVD Subtypes & Applications
Two main PVD methods serve different applications:
Thermal evaporation vaporizes material through heating, making it the standard choice for metallizing plastics and decorative finishes on non-conductive substrates like cosmetic packaging components.
Magnetron sputtering produces harder functional coatings—TiN, ZrN, and TiCN—built for industrial performance. TiAlN coatings reach 33 GPa hardness, well above what electroplated alternatives can achieve.
Key Performance Benefits
- TiN coatings achieve 2,500 HV (30 GPa) versus 940–1,210 HV for hard chrome — a meaningful hardness advantage
- Molecular bonding resists peeling and chipping where surface deposits fail
- Vacuum deposition ensures consistent color uniformity across the entire part
- Parts emerge ready for assembly with no post-processing required
- Zero toxic disposal: RoHS-compliant with no hazardous chemical byproducts
Use Cases of PVD
PVD excels in applications requiring dimensional precision and substrate versatility:
- Cosmetic packaging: metallic finishes on plastic caps, compacts, and fragrance bottles
- Automotive trim: durable finishes that hold tight tolerances
- Medical instruments: biocompatible coatings with superior wear resistance
- Industrial tools: TiN-coated forming tools show 4x service life improvement
PVD applies to broader substrate ranges than electroplating—including engineering-grade plastics like Nylon, Ultem, and LCP—enabling finish matching between metal and plastic components on the same product. This matters most in premium cosmetics and consumer goods, where visual consistency across mixed materials is non-negotiable.
Providence Metallizing introduced magnetron sputtering in the 1980s, building decades of process knowledge across both decorative and industrial PVD applications.
What is Electroplating?
Electroplating is a process where electric current drives metal ions from a chemical solution onto a conductive substrate, depositing a metallic layer. With over a century of application history, it's one of the most widely used industrial finishing methods across manufacturing sectors.
The Electroplating Process
The part acts as the cathode, attracting positive metal ions from the anode material dissolved in the bath. As current flows, metal deposits onto the surface — and deposit thickness increases with time in the solution. Common coating metals include gold, silver, nickel, chrome, and copper.
Process Limitations
Electroplating operates as a low-energy process—ions arrive with limited kinetic energy, resulting in weaker adhesion compared to PVD. Edge build-up occurs because electricity travels the path of least resistance, creating thicker deposits on prominent edges and thinner coverage in recesses.
Substrate Restrictions: Certain metals cannot be electroplated directly:
- Aluminum: zincate treatment (ASTM B253) replaces the natural oxide layer before plating
- Titanium: requires harsh activation using hydrofluoric acid-formamide solutions
- Magnesium: needs electroless nickel pre-plating (ASTM B480) first
- Non-conductive polymers: require multi-step pre-treatment including chromic acid etching and palladium catalyzation
Core Advantages
Despite its limitations, electroplating offers practical advantages that keep it relevant across many applications:
- Lower equipment investment than PVD or CVD systems
- Effective precious metal deposition (gold, silver, rhodium)
- Handles large, complex shapes with relative ease
- Flexible color and finish adjustment simply by changing the bath chemistry
- Deep process knowledge available across virtually every manufacturing industry
Use Cases of Electroplating
Electroplating fits applications where precious metals or established decorative finishes are priorities:
- Decorative jewelry finishes
- Gold/silver plating for tableware
- Chrome for automotive and plumbing components
- Nickel for corrosion protection (1.5–2.5 microns typical)
It performs best on conductive substrates like brass, copper, and steel — though with the right pre-treatment, it can also be applied to stainless steel and plastics.
PVD vs. Electroplating: Which is Right for Your Application?
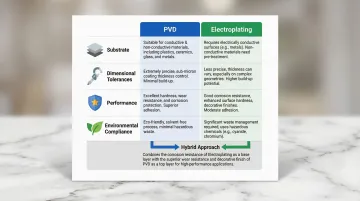
Key Decision Factors
| Factor | Choose PVD | Choose Electroplating |
|---|---|---|
| Substrate | Plastics, engineering polymers, reactive metals (titanium, aluminum) | Standard conductive metals (brass, copper, steel) |
| Dimensional Tolerances | Ultra-thin coatings (2–5 µm) preserve razor edges; no post-machining | Hard chrome over 50 µm can exaggerate imperfections; often requires finish grinding |
| Performance | Superior hardness and wear resistance for functional applications | Flexible decorative finishes; precious metal aesthetics |
| Environmental | RoHS and REACH compliant; zero emissions | Requires wastewater treatment and strict EPA compliance |
Situational Guidance
Choose PVD when:
- Dimensional precision is critical
- Substrates include plastics or hard metals
- Finish consistency across high volumes is required
- Environmental compliance limits chemical use
- Superior hardness and wear resistance matter
Choose Electroplating when:
- Precious metal deposition is needed
- Substrate is standard conductive metal
- Decorative flexibility matters more than hardness
- Lower initial capital investment is priority
The Hybrid Approach
Not every application fits cleanly into one category. PVD can serve as a base adhesion layer before electroplating, which is useful for substrates that don't etch well with standard electroplating alone. The result: PVD's adhesion strength paired with electroplating's precious metal aesthetics.
Finish-Matching Challenge
Products combining metal and plastic parts — common in cosmetics packaging, consumer electronics, and hardware — often require both methods to achieve visual consistency. A supplier capable of running both processes in-house becomes critical for this.
Providence Metallizing's 60+ years working with both PVD and electroplating enables finish-matching across substrates for major beauty brands including Estée Lauder, L'Oreal, Avon, and Elizabeth Arden.
Real-World Applications: Where Each Method Wins
Cosmetics Packaging Multi-Component Assembly
A cosmetics manufacturer needs premium metallic finishes across a fragrance cap (metal) and collar (plastic). Electroplating alone cannot coat the plastic component, but PVD handles the plastic while electroplating addresses the metal—both dialed in to match visually.
Providence Metallizing has solved this challenge for major beauty brands by leveraging dual finishing capabilities. Their 2011 Finish of the Year recognition for the Viva La Juicy bottle demonstrates how sophisticated multi-process finishing creates cohesive aesthetics across mixed materials.
Industrial Tooling Performance
For injection molds and cutting tools, PVD coatings outperform electroplating because tight tolerances must be maintained. The performance gains are measurable:
- AlCrN coated carbide tools outperform TiN coated tools by nearly 70% under high cutting speeds
- PVD diffusion treatments raise injection mold surface hardness to 1,400 HV, replacing hard chrome without edge embrittlement
Providence Metallizing offers free sampling for most applications, letting production teams confirm finish quality and substrate compatibility before committing to full runs. With over 60 years of experience and both PVD and electroplating capabilities in-house, they can match the right process to each material and volume requirement.
Conclusion
PVD outperforms electroplating in durability, substrate versatility, environmental compliance, and tight tolerance preservation. Electroplating remains relevant for precious metal finishes, certain decorative applications, and cost-sensitive situations. The best choice depends on what the part needs to do, what it's made of, and where it will be used.
These trade-offs matter most for manufacturers producing high-volume, multi-component products — particularly in cosmetics, automotive, and electronics — where finish consistency and total production cost are directly tied to process selection. Partnering with a finisher experienced in both processes, like Providence Metallizing Company, gives you the flexibility to evaluate options side by side and choose based on actual part requirements rather than process familiarity alone.
Frequently Asked Questions
Is PVD coating better than electroplating?
PVD generally outperforms electroplating in durability, adhesion, and environmental profile, but electroplating remains preferred for precious metal deposition and certain decorative applications. "Better" depends on your specific substrate, performance requirements, and whether you need functional wear resistance or decorative precious metal finishes.
What metals cannot be electroplated?
Titanium, aluminum, and magnesium are difficult to electroplate directly because their natural oxide layers require harsh pre-treatments to achieve adhesion. Non-conductive materials like plastics also require multi-step conductive pre-treatment before electroplating, whereas PVD handles these substrates without that added complexity.
Does PVD plating wear off?
PVD coatings resist wear and do not peel like electroplated coatings can, but they can gradually thin over years of extreme abrasion. With proper substrate preparation, PVD finishes on cutting tools increase service life by 300% to 1000% depending on application.
Can PVD and electroplating be used together?
Yes, PVD can be applied as a base adhesion layer before electroplating, which is useful for substrates that don't bond well with electroplating alone. The combination pairs PVD's strong adhesion with electroplating's ability to deposit precious metal decorative finishes.
Which process is more environmentally friendly?
PVD is the cleaner process since it takes place in a vacuum with no chemical baths, producing no hazardous wastewater. Electroplating requires chemical management and wastewater treatment per EPA regulations (40 CFR 413/433), adding cost and regulatory overhead—especially relevant for RoHS-compliant manufacturing environments.
How thick is a PVD coating compared to electroplating?
PVD coatings typically range from 2–5 microns for functional applications, thinner than many electroplated deposits. The atomic-level bonding makes PVD coatings more durable per unit thickness, and the thinness is an advantage when dimensional tolerances must be maintained without post-machining.


