
Introduction
Chrome-finished plastic is now standard across automotive trim, cosmetic packaging, consumer electronics, and hardware. But plastic cannot be chrome plated the same way metal can. Unlike conductive metal substrates, plastic requires extensive chemical and electrochemical preparation before the first metal layer can be deposited — making this a multi-step process where precision matters at every stage.
Results vary dramatically based on plastic type, part geometry, and process chemistry. A properly plated part demands tight control over etching parameters, activation chemistry, bath temperatures, and current density. Minor deviations at any stage — under-etching the substrate or contamination before activation — cascade into visible defects like peeling, pitting, or dull spots.
That chain of dependencies is what makes this process unforgiving.
This guide walks you through the complete step-by-step chrome plating process for plastic, explains which plastic types are compatible (and which aren't), identifies the key variables that control finish quality, highlights common mistakes, and explores when alternatives like PVD or vacuum metallizing may be more practical.
TL;DR
- Chrome plating plastic involves six steps: cleaning, etching, electroless coating, copper buildup, nickel, then chrome
- ABS is the most compatible plastic; polyethylene and polypropylene are generally incompatible with chrome plating
- Sharp edges, deep recesses, and uneven wall thickness cause predictable plating defects — part design matters
- Bath temperature, current density, etching chemistry, and layer thickness all determine finish quality
- Professional plating services deliver more consistent results than DIY for high-volume or cosmetic-grade parts
How to Chrome Plate Plastic: Step-by-Step Process
Chrome plating plastic is fundamentally different from plating metal. The process involves multiple chemical and electrochemical steps to transform a non-conductive plastic surface into a brilliant, durable chrome finish.
Step 1: Clean and Prepare the Plastic Part
Thoroughly degrease the plastic surface to remove mold release agents, oils, fingerprints, and contaminants. Any residue left at this stage prevents metal adhesion and causes blistering or peeling in the final finish. Even microscopic contamination creates isolated areas where no metal bonds to the substrate.
Inspect plastic parts for surface defects before processing begins:
- Sink marks from uneven cooling during molding
- Weld lines where plastic flows meet during injection
- Scratches, scuffs, or machining marks
- Surface porosity or texture irregularities
Chrome plating amplifies surface imperfections rather than hiding them. A defect invisible on bare plastic becomes glaringly obvious under a reflective chrome finish.
Critical requirement: Only rigid plastic parts should enter the chrome plating process. Flexible or semi-flexible substrates will crack under thermal and chemical stress during subsequent steps. Materials like thermoplastic polyurethane (TPU), flexible polyethylene, and soft polypropylene grades are incompatible with traditional chrome plating.
Step 2: Etch the Plastic Surface
Chemical etching micro-roughens the plastic surface to create mechanical bonding sites for metal layers. For ABS (the most commonly plated plastic), etching uses a chromic acid and sulfuric acid solution with carefully controlled concentration and temperature.
Standard etching parameters for ABS:
- Chromic acid (CrO₃) concentration: 350–430 g/L
- Sulfuric acid (H₂SO₄) concentration: 380–400 g/L
- Operating temperature: 60°C to 70°C
- Dwell time: 5 to 15 minutes (typically 8–12 minutes for small parts, 15–25 minutes for large/complex geometries)
During etching, the sulfochromic acid selectively oxidizes and dissolves the polybutadiene (PBD) rubber phase spheres distributed within the styrene-acrylonitrile (SAN) matrix. This creates interconnected micro-holes or "anchor holes" typically 1–5 µm in size that provide mechanical interlocking sites for subsequent metal layers.
Etching is the most chemically sensitive step in the entire process:
- Under-etching creates insufficient pore formation, leading to poor adhesion and peeling
- Over-etching degrades polymer strength, forms internal voids that act as stress concentrators, reduces peel force necessary for delamination, and produces a dull surface appearance

Step 3: Apply an Electroless Conductive Layer
Plastic is non-conductive, so electricity cannot drive metal deposition directly onto the bare surface. An electroless (chemical) deposition of copper or nickel must be applied first. This layer deposits uniformly across the entire surface without requiring electrical current.
Activation sequence before electroless plating:
The substrate must be catalyzed to initiate autocatalytic metal deposition. The standard two-step activation process involves:
- Sensitization: Immersion in acidic stannous chloride (SnCl₂) solution
- Activation: Immersion in palladium chloride (PdCl₂) solution
The Sn(II) ions adsorb onto the etched substrate and reduce Pd(II) ions to Pd(0), forming palladium nuclei that catalyze electroless metal deposition.
Electroless plating thickness requirements:
- Electroless copper: minimum 1.0 µm (40 micro-inches)
- Electroless nickel: 0.25 µm to 1.5 µm (10 to 60 micro-inches), depending on application
The quality of this conductive layer is the foundation of the entire process. Thin spots, gaps, or poor adhesion propagate as defects through every subsequent layer. No amount of additional plating can compensate for a defective electroless base.
Step 4: Electroplate with Copper and Nickel Base Layers
With the conductive surface established, the part is submerged in electroplating baths where electrical current drives metal deposition onto the substrate. Two base layers are built up in sequence before chrome is applied:
- Copper goes down first to build thickness and fill minor surface irregularities, creating a smooth, uniform base for subsequent layers.
- Bright nickel is applied over copper to establish the reflective base that makes chrome appear brilliant. The bright nickel layer—not the chrome itself—is primarily responsible for the mirror-like appearance of decorative chrome. Standard operating temperatures for bright nickel decorative plating range from 52°C to 62°C.
Chrome itself is a thin top layer that protects the nickel and adds its characteristic blue-white tint. Without a high-quality bright nickel underlayer, chrome appears dull and grey rather than brilliant and reflective.

Step 5: Apply the Final Chrome Layer
The final electroplating step deposits chromium from either a trivalent or hexavalent chromium solution. For decorative applications, chrome thickness typically ranges from 0.05 to 0.5 µm, with 0.1 to 0.3 µm common for automotive and consumer goods.
Trivalent vs. hexavalent chromium:
Trivalent chromium baths are increasingly preferred over hexavalent chromium due to:
- Lower toxicity and reduced health hazards
- Easier regulatory compliance (RoHS directive restricts hexavalent chromium to maximum 0.1% by weight)
- Less hazardous waste generation
- Superior throwing power and covering power compared to hexavalent baths
While hexavalent chrome historically produced slightly brighter results, modern trivalent processes have narrowed this gap significantly. Standard operating temperatures for decorative hexavalent chrome range from 35°C to 45°C, while trivalent chrome operates between 30°C and 55°C depending on formulation.
After chrome deposition, parts are rinsed thoroughly, inspected for coverage and brightness uniformity, and dried. Any areas with dull spots, pitting, or incomplete coverage typically trace back to failures in earlier steps—contamination during cleaning, uneven etching, gaps in the electroless layer, or poor current distribution during electroplating.
Which Plastics Can Be Chrome Plated (and Which Cannot)
Not all plastics are suitable for chrome plating. The substrate must withstand chemical etching, accept metal adhesion, and remain dimensionally stable under thermal and chemical stress.
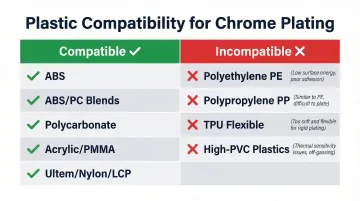
ABS: The Gold Standard for Chrome Plating
ABS (Acrylonitrile Butadiene Styrene) is the most commonly and successfully chrome plated plastic. Its butadiene rubber phase is selectively etched by chromic acid, creating ideal mechanical anchoring sites for metal layers. The selective dissolution of butadiene spheres creates the interconnected micro-pore structure that enables superior metal adhesion.
Plastics That Can Be Chrome Plated with Process Adaptation
Several other plastics can be chrome plated when using modified etching chemistry or additional process steps:
- ABS/PC blends: Require a "sweller" or solvent conditioning step before chromic acid etching to increase etch penetration and uniformity, compensating for polycarbonate's higher chemical resistance
- Polycarbonate (PC): Plateable with specialized pretreatment; results are sensitive to molecular weight and part wall thickness
- Acrylic (PMMA): Compatible with adapted processes, though surface crazing during etching is the primary risk to control
- Engineering grades: Select materials such as Ultem (PEI), Nylon (PA), and LCP (Liquid Crystal Polymer) can be plated, though most standard plating operations are not equipped to handle these substrates — Providence Metallizing's plating-on-plastic line is purpose-built for these materials.
Plastics That Cannot Be Chrome Plated
Flexible materials and commodity resins consistently fail — either the surface won't etch properly, or the part deforms before plating is complete:
- Polyethylene (PE): Does not etch well, resists metal adhesion, deforms under process conditions
- Polypropylene (PP): Standard grades are incompatible; specialized pretreatment is required for any success
- Thermoplastic Polyurethane (TPU): Flexible substrate physically deforms under chemical and thermal stress
- High-PVC content plastics: Resist necessary surface modification and fail metallic adhesion tests
When a substrate can't hold its shape or accept an etch, no amount of process adjustment produces a durable finish — parts end up rejected at adhesion testing.
Key Parameters That Affect Chrome Plating Results on Plastic
The difference between a flawless chrome finish and a batch of rejected parts almost always comes down to tight control of a handful of process variables. The difference between a flawless chrome finish and a batch of rejected parts almost always comes down to tight control of a handful of process variables. Four factors drive most outcomes: etch chemistry, bath temperature, current density, and part design.
Etching Chemistry and Time
The etch step defines how well metal layers mechanically bond to the plastic — incorrect concentration or dwell time is the most common cause of adhesion failure. Authoritative industry guidelines specify the following operating parameters:
- Chromic acid concentration: 350–430 g/L
- Sulfuric acid concentration: 380–400 g/L
- Temperature: 60°C to 70°C
- Dwell time: 5 to 15 minutes (varies by part size and complexity)
Under-etching produces insufficient pore formation and local missing plating. Over-etching degrades polymer strength, creates internal voids, reduces peel resistance, and causes dull appearance.
Bath Temperature
Bath temperature directly affects current distribution, deposit uniformity, and chrome layer brightness. Too low and coverage suffers; too high and the plastic substrate can warp. Temperature deviations appear as finish inconsistencies:
- Bright nickel baths: 52°C to 62°C standard range
- Hexavalent chrome baths: 35°C to 45°C typical
- Trivalent chrome baths: 30°C to 55°C depending on formulation
Operating outside these ranges produces uneven deposits, reduced brightness, or thermal distortion of plastic parts.
Current Density
Current density controls deposition rate and determines whether chrome deposits uniformly or concentrates on edges and high points. Hexavalent chromium baths suffer from low throwing power due to poor cathode efficiency (10–20%), resulting in excessive build-up on sharp edges and inadequate coverage in recessed areas.
Complex geometries with recesses or sharp corners are hardest to plate uniformly. Trivalent chromium baths offer notably better throwing power compared to hexavalent baths, distributing chrome more evenly across complex surfaces.
For difficult geometries, auxiliary or conforming anodes direct current into low current density areas and prevent over-plating on prominent edges. NASF guidance recommends using separate power supplies for auxiliary anodes to strictly control current distribution.
Part Design and Wall Thickness
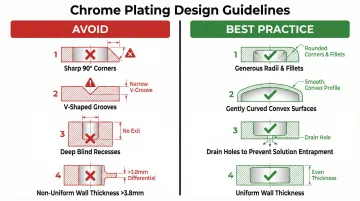
Molding quality and part design directly influence plating outcomes. Wall thickness variations cause uneven cooling during molding, building residual stress that causes plated layers to crack or delaminate.
Design guidelines specify wall thicknesses should not exceed 3.8mm (0.150 inches) to avoid uneven cooling, shrinkage, and warping.
Problematic features that routinely cause plating defects:
- Sharp 90-degree angles: Cause excessive plate build-up (flash) due to high current density
- V-shaped grooves: Trap plating solutions (causing cross-contamination) and fail to plate adequately without auxiliary anodes
- Deep recesses: Difficult to rinse, prone to solution entrapment, receive insufficient current
- Raised or depressed lettering: Creates uneven current distribution
Design best practices:
- Incorporate gently curved/convex surfaces
- Use generous radii and fillets instead of sharp transitions
- Add drain holes to prevent solution entrapment
- Avoid deep blind holes or sharp internal corners
Common Mistakes When Chrome Plating Plastic
Skipping or Rushing Surface Preparation
Contamination from mold release agents, fingerprints, or machining oils that isn't fully removed before etching creates isolated areas where no metal adhesion occurs. These show up as blistering, flaking, or bare spots after plating. A thorough cleaning and activation sequence before etching is what determines whether the plated finish holds — or fails within weeks.
Selecting the Wrong Plastic or Processing a Flexible Substrate
Attempting to chrome plate polyethylene, polypropylene, or any semi-flexible resin without confirming material compatibility first leads to adhesion failure. The etchant cannot create adequate bonding sites in these materials, and mechanical stress from thermal cycling causes delamination. Always verify substrate compatibility before committing to tooling and production.
Ignoring Part Design Constraints Before Plating
Submitting parts with sharp internal angles, deep blind recesses, or non-uniform wall thickness without design review results in predictable defects:
- Plate buildup on edges
- Bare or poorly plated recesses
- Stress cracking from residual molding stress
These defects cannot be fixed after the fact. Design review before tooling is cut is far cheaper than rework or scrap.
Alternatives to Traditional Chrome Plating on Plastic
Traditional electroplating on plastic produces the most durable and authentic chrome finish, but other methods exist for applications where the full process is not practical or necessary.
Physical Vapor Deposition (PVD)
PVD suits applications requiring environmental compliance, thinner substrates, or greater design flexibility. PVD processes operate at maximum temperatures of 80°C to 100°C, safely below the thermal deformation thresholds of heat-sensitive plastics like ABS and PC. The process uses no harsh acid baths and deposits chrome-like finishes on a wider range of plastic types, including those that cannot be chemically etched.
The trade-off is cost. Smaller batch coaters range from $500,000 to $800,000, while fully automated cluster lines cost between $3,000,000 and $5,000,000. This makes PVD practical only for high-volume production.
The resulting finish, while visually similar, is thinner than electroplated chrome. PVD coatings do achieve extreme hardness values of 1500 to 3000 HV, compared to 500 to 1100 HV for electroplated decorative chromium.

Vacuum Metalizing (Aluminum Metallizing)
Cosmetic packaging and reflector manufacturers favor vacuum metalizing when a chrome-like appearance is needed at lower cost. Vacuum metalizing deposits aluminum (not chromium) in a vacuum chamber over a basecoat, creating a reflective surface.
The finish is not true chrome and lacks the corrosion and scratch resistance of electroplated chrome. Protective topcoats are required, and the finish can appear silver rather than chrome in some lighting conditions. The aluminum layer is fragile — if the topcoat chips, the aluminum oxidizes rapidly and loses adhesion. Electroplated chrome and PVD chrome offer significantly better corrosion and scratch resistance.
Chrome Spray Paint / Chrome Film
These options work only for decorative or prototype applications where appearance is the sole goal and no durability is required. Chrome spray and chrome film require no industrial equipment, which keeps entry costs low.
That convenience comes at a steep quality cost:
- Chrome spray paint reads more silver than chrome and offers no meaningful durability
- Film laminates wrinkle, tear, and peel over time
- Neither method replicates true chrome in feel, depth, or longevity
- Both are unsuitable for any production part
Frequently Asked Questions
Can plastics be chrome plated?
Yes, certain plastics—particularly ABS and ABS/PC blends—can be chrome plated through a multi-step process that first makes the surface conductive via electroless plating. Flexible plastics and many commodity resins like polyethylene and standard polypropylene cannot be successfully plated using traditional methods.
What is the average cost for chrome plating?
Chrome plating costs on plastic vary widely based on part size, geometry complexity, layer requirements, and volume. Electroless nickel plating (the foundational conductive layer) typically costs between $7 and $20 per square foot, with total costs rising based on labor, energy, waste treatment, and chemical replenishment. Requesting sample runs from a plating service before committing to full production is the most reliable way to assess feasibility and cost.
How long does chromium plating last?
Decorative chrome plating on plastic can last many years in indoor applications. Properly specified multi-layer copper/nickel/chromium electroplate provides excellent durability for both indoor and severe outdoor/UV-exposed applications like automotive exterior trim. Outdoor durability depends on UV exposure, temperature cycling, and topcoat protection. Industry standards like ASTM B368 (CASS testing) and ISO 4525 define performance requirements for various service conditions.
Where is chromium plating done?
Chrome plating on plastic is performed by specialized electroplating facilities that maintain chemical baths, waste treatment systems, and regulatory compliance. It cannot be done at home due to hazardous chemical handling requirements and complex process controls. Look for facilities with demonstrated experience on your specific substrate, particularly if you're working with engineering-grade plastics like Ultem, Nylon, or LCP.
Does chrome plating use chromium?
Yes, chrome plating deposits actual chromium metal onto the surface—either from a hexavalent chromium bath (being phased out due to toxicity and REACH Annex XIV restrictions) or a trivalent chromium bath (increasingly the industry standard). The term "chrome" in spray paints or films refers only to the appearance—not actual chromium content.


