
Plastic electroplating solves this by depositing a real metal layer onto plastic surfaces. Unlike spray-on metallic paints, electroplating creates a true metallized finish with corrosion resistance, wear durability, and brilliant reflective surfaces. The global plating on plastics market reached $614.5 million in 2023 and is projected to grow to $1.01 billion by 2030, expanding at a 7.4% CAGR according to Grand View Research.
This guide covers everything you need to understand plastic electroplating: what it is, why plastic requires special preparation, the complete step-by-step process, and the four main classification methods used across industries.
TLDR
- Plastic electroplating coats non-conductive plastic with metal by first making the surface conductive through chemical pre-treatment
- Key steps: cleaning, chemical etching (microscopic anchor points), surface catalyzation, electroless plating for conductivity, then standard electroplating
- Four main methods: electroless (autocatalytic) plating, wet electroplating, vacuum/PVD plating, and electroforming
- Delivers 20–40% weight savings over solid metal while producing jewelry-grade metallic finishes
- Widely used in automotive, consumer electronics, cosmetic packaging, and medical device industries
What Is Plastic Electroplating?
Electroplating is an electrochemical process that uses electric current to deposit a thin, uniform layer of metal onto a surface. The workpiece acts as the cathode (negative), a metal source acts as the anode (positive), and dissolved metal ions in an electrolyte solution are driven onto the part by the current.
Plastic electroplating differs from metal electroplating in one critical way: plastics are non-conductive, so they cannot take part in the electrochemical reaction. That non-conductivity makes plastic electroplating a multi-stage process — specialized pre-treatment is required before any metal deposition can begin.
Why Electroplate Plastic?
The primary purposes include:
- Corrosion and wear resistance — metal coatings protect plastic substrates from environmental degradation and abrasion
- Enhanced aesthetics — achieve brilliant chrome, gold, brushed metal, or matte finishes at scale
- Adds conductivity for EMI/RFI shielding, or creates insulating barrier coatings where needed
- Delivers metal-like appearance and protection at a fraction of solid metal's weight
- Cost efficiency — luxury-grade finishes at far lower cost than solid metal components
For industries like cosmetic packaging, achieving jewelry-level metallic finishes on plastic at high volumes is a core manufacturing requirement — one that Providence Metallizing Company has served for over 25 years as an approved supplier to brands including Estée Lauder, L'Oreal, and Avon.
Why Plastic Requires Special Preparation Before Electroplating
The Conductivity Barrier
Standard electroplating depends entirely on substrate conductivity. Plastics (including ABS, polycarbonate, nylon, and engineering polymers) lack this property. To make electroplating possible, the plastic surface must be mechanically and chemically modified so a conductive base layer can be deposited.
Not All Plastics Are Created Equal
ABS is the most commonly electroplated plastic. Its butadiene component responds predictably to chemical etching: strong oxidizing agents selectively remove the butadiene phase, creating microscopic pores that act as mechanical bonding sites for metal deposits. Other plastics are plateable too, but each demands a different pre-treatment approach:
| Plastic | Plateability | Pre-treatment Requirements |
|---|---|---|
| ABS/PC Blends | Widely plateable; higher heat resistance than ABS | Optimized etching parameters (higher temperatures or specific acid concentrations) |
| Polypropylene (PP) | Plateable; excellent chemical resistance | Aggressive etching, plasma, or corona treatments due to lack of double-bonded carbons |
| Nylon (PA) | Plateable for selected grades | Proprietary pre-treatments like Atotech Adhemax PA |
| Ultem (PEI) | Plateable; high heat resistance (170°C RTI) | Can be metallized without primer; often uses permanganate etch to open imide ring |
| Liquid Crystal Polymer (LCP) | Plateable; low dielectric constant | Requires alkaline pre-etching or plasma treatment (N2) to introduce hydrophilic polar groups |

Design Considerations That Affect Platability
Poor part design is one of the leading causes of adhesion failure in plastic electroplating. Critical design factors include:
- Uniform wall thickness — Avoid walls thicker than 6.35 mm (0.250 inches) to prevent sinks and voids
- Draft angles — Minimum 1° draft facilitates mold release without contaminating release agents
- Radii and fillets — Avoid sharp corners; prefer 1/16 inch minimum for inside angles, 1/32 inch for external angles
- Blind holes — Trap plating solutions and cause cross-contamination; if necessary, limit depth to half the diameter
- Gate placement — Poor gate location creates stress concentrations that magnify under bright metal plating
The Plastic Electroplating Process: Step by Step
Step 1 — Cleaning and Degreasing
The plastic part must be thoroughly cleaned to remove mold release agents, oils, fingerprints, and contaminants. Alkaline or solvent-based cleaning agents are used, followed by water rinsing.
Incomplete cleaning causes adhesion failure. No subsequent step can compensate for contamination left at this stage.
Step 2 — Etching (Chemical Roughening)
The cleaned plastic is immersed in a chromic acid-sulfuric acid etch bath (or chromium-free alternative) that chemically attacks the butadiene phase of ABS. This creates microscopic pores and anchor points on the surface.
The metal-to-plastic bond relies on mechanical interlocking, not chemical bonding. The etched surface provides the physical structure necessary for durable adhesion.
Traditional chemistry runs at 350–425 g/L CrO₃ and 180–400 g/L H₂SO₄ at 50–70°C. Regulatory restrictions on hexavalent chromium (Cr(VI)) are expected to take effect in Europe between late 2027 and early 2028. As a result, manufacturers are adopting permanganate-based etches or proprietary alternatives such as MacDermid Enthone's evolve and Atotech's Covertron 600.
Sandblasting is a non-chemical alternative, providing mechanical roughening without an etch bath.
Step 3 — Sensitization and Activation (Catalyzing)
This two-stage process first sensitizes the etched surface with stannous chloride (SnCl₂), then activates it with palladium chloride (PdCl₂). The palladium deposits a thin catalytic layer that will initiate the next chemical reduction step.
Single-step colloidal suspensions of pre-reduced palladium/tin particles are a modern alternative that streamlines this two-stage sequence.
This step activates the plastic surface for metal deposition. Without proper catalyzation, electroless plating cannot proceed.
Step 4 — Electroless Plating (Base Metal Layer)
The activated plastic is immersed in an electroless copper or electroless nickel bath. Metal ions in the solution are chemically reduced — without external current — and deposited uniformly across the catalyzed surface.
Thickness: Typically 0.5 to 1 micron for the base coat, though EMI shielding applications may require 1.0 µm to 10.0 µm of electroless copper.
The result is a uniformly conductive surface — the essential bridge between a non-conductive plastic substrate and the electroplating baths that follow.
Step 5 — Electroplating (Build-Up and Finish Layers)
Once conductive, the part enters standard electroplating baths:
- Copper plating — Builds thickness (12–25 µm) and smooths the micro-roughened surface
- Nickel plating — Adds corrosion resistance (typically 15–30 µm) and serves as a base for decorative layers
- Final finish — Chrome, gold, nickel, or other metals depending on application

ISO 4525 specifies grades up to 15 µm copper + 30 µm nickel + 0.3 µm chrome for high-durability decorative applications.
Classification of Plastic Electroplating Methods
Method 1 — Electroless Plating (Autocatalytic Plating)
Electroless plating deposits metal without any external electrical current. Metal ions in solution are reduced by a chemical reducing agent onto a catalyzed surface — making it the standard pre-treatment base layer in plastic electroplating (Step 4), and a standalone solution for EMI shielding and solderability on electronics enclosures.
Its defining advantage: uniform deposition across complex geometries, including internal cavities and blind holes. In shielding applications, 1.0 µm electroless copper + 0.25 µm electroless nickel routinely achieves 80–100 dB shielding effectiveness — meeting FCC Class B and MIL-STD-461 requirements.
Method 2 — Wet Electroplating (Standard Galvanic Electroplating)
The traditional electrolytic process: external DC current drives metal ions from an anode through an electrolytic solution onto the cathode (the part). On plastic, it always follows an electroless base coat.
Wet electroplating is the most widely used method for high-volume decorative parts — particularly in automotive and cosmetic packaging — because it offers:
- Wide metal selection (copper, nickel, chrome, gold, tin)
- Precise thickness control, up to 60 µm
- Lower per-unit cost at scale
- Decorative and functional finish options in a single process
Method 3 — Vacuum Plating (PVD/Sputtering)
A dry process conducted in a vacuum chamber: metal or alloy is vaporized — via thermal evaporation, magnetron sputtering, or ion plating — and deposited as an ultra-thin film directly onto the plastic surface.
Advantages over wet electroplating include:
- Cycle times as short as 25 minutes (vs. 2.7 hours for wet processes)
- Cr(VI)-free process with no heavy metal wastewater
- Broad substrate compatibility, including PC, PA, TPE, and non-platable grades
- Wide material options: TiN, ZrN, and sputtered metals
The trade-off is thinner coatings (0.2–0.5 µm vs. up to 60 µm for wet plating) and higher equipment costs. PVD is the go-to for high-end decorative products, automotive interiors, consumer electronics, and EMI/RFI shielding.

Method 4 — Electroforming
Electroforming uses electrolytic deposition to build thick metal structures on a mandrel or mold form, which is then separated from the deposited metal. It's not a direct coating technique for plastic — instead, it produces mold inserts that impart special surface textures (sharp polished or etched boundary lines) to injection-molded parts. The result: premium surface effects that standard electroplating cannot replicate.
Method 5 — Conductive Paint
Rather than electroless plating, conductive paint — loaded with copper, silver, or nickel particles — is sprayed or dipped onto plastic to create a conductive surface before electroplating begins.
Setup is faster and initial costs are lower, but there are real trade-offs:
- Adhesion is less durable than a full electroless process
- Line-of-sight application creates coverage gaps on complex geometries
- High pigment concentrations are required to maintain electrical continuity
Best suited for prototypes, small runs, or parts where cosmetic quality requirements are less demanding.
Benefits and Industrial Applications of Electroplated Plastic
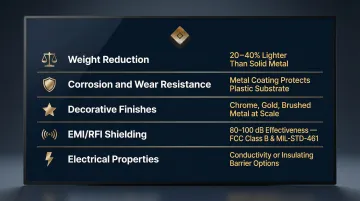
Functional and Aesthetic Benefits
- Cuts weight by 20-40% versus steel — one automotive front-end module dropped from 13.43 kg to 5.65 kg (58%) after switching to plated reinforced polypropylene
- Shields against corrosion and wear through metal coatings that protect the underlying plastic from environmental degradation and abrasion
- Delivers chrome, gold, brushed metal, and matte finishes at scale — luxury-grade aesthetics at a fraction of solid metal cost
- Provides 80-100 dB EMI/RFI shielding effectiveness via electroless copper/nickel stacks, meeting FCC Class B and MIL-STD-461 requirements
- Adds targeted electrical properties — either conductivity for ESD control or insulating barrier coatings, depending on the application
Key Industries and Applications
| Industry | Common Applications |
|---|---|
| Automotive | Grilles, light bezels, mirror housings, door handles, interior trim |
| Consumer Electronics | Smartphone frames, smartwatch cases, camera deco rings, wearable housings (EMI shielding) |
| Cosmetic & Personal Care | Perfume caps, lipstick tubes, compact cases, pump dispensers |
| Plumbing & Hardware | Faucet handles, showerheads, bathroom fittings |
| Medical Devices | Equipment housings requiring EMI/RFI shielding for sensitive electronics |
| Writing Instruments & Tableware | Decorative and functional metal finishes |
Cosmetic packaging carries the strictest quality standards of any sector. Quality control follows ISO 2859-1 sampling plans with defined Acceptable Quality Limits: 0% AQL for critical defects, 1.5% for major defects, and 2.5% for minor defects.
Providence Metallizing Company: Specialized Expertise
Providence Metallizing Company stands as a domestic leader in plating-on-plastic for the cosmetic industry, with over 60 years of metallizing experience and more than 25 years serving cosmetic and personal care markets. The company offers over 30 different finishes on plastic substrates, including chrome, gold, silver, brushed stainless, hematite, architectural finishes, and satin nickel.
Key capabilities:
- Plating on engineering-grade plastics including Ultem, Nylon, and LCP
- Free sampling services for most applications
- High-volume production capacity: over one million parts per week
- 24/5 production schedule with half the normal lead times of standard finishing operations
Approved supplier status with Estée Lauder, L'Oreal, Avon, and Elizabeth Arden reflects the consistency that high-volume cosmetic production demands — and what a purpose-built plating-on-plastic operation can reliably deliver.
Frequently Asked Questions
What is the process of plastic electroplating?
Plastic electroplating involves cleaning the part, etching the surface to create microscopic anchor points, catalyzing with palladium, depositing an electroless copper or nickel base coat to create conductivity, then building up layers of copper, nickel, and decorative chrome or gold through standard electroplating.
What are the steps in electroplating?
The core steps are: surface preparation (cleaning and chemical etching), palladium catalyzing, and electroless deposition of a conductive base layer (0.5–1 µm copper or nickel). Electrolytic plating then builds up copper (12–25 µm), corrosion-resistant nickel (15–30 µm), and a decorative top coat of chrome, gold, or other finish.
What are the 4 methods of plating?
The four main plating methods are:
- Electroless (autocatalytic) — chemical reduction deposits metal without external current
- Wet (galvanic) electroplating — electric current drives metal ion deposition
- Vacuum/PVD plating — metal is vaporized in a vacuum chamber and condenses onto the substrate
- Electroforming — builds thick, freestanding metal parts on a removable mandrel
What plastics can be electroplated?
ABS is the most commonly plated plastic due to its favorable response to chemical etching. ABS/PC blends, nylon, and polypropylene can also be plated. Engineering grades like Ultem and LCP are plateable but require specialized process expertise and proprietary pre-treatments.
What are the main benefits of electroplating plastic?
Electroplating plastic delivers corrosion and wear resistance, brilliant metallic finishes, and 20–40% weight savings versus solid metal. It also provides EMI/RFI shielding for electronics and keeps per-part costs competitive in high-volume production.
How durable is electroplated plastic compared to solid metal parts?
Electroplated plastic offers strong surface durability through the metal coating, though the plastic substrate cannot match the structural strength of solid metal. Adhesion quality and coating thickness are the key variables — parts designed and processed correctly perform well across most demanding environments.


